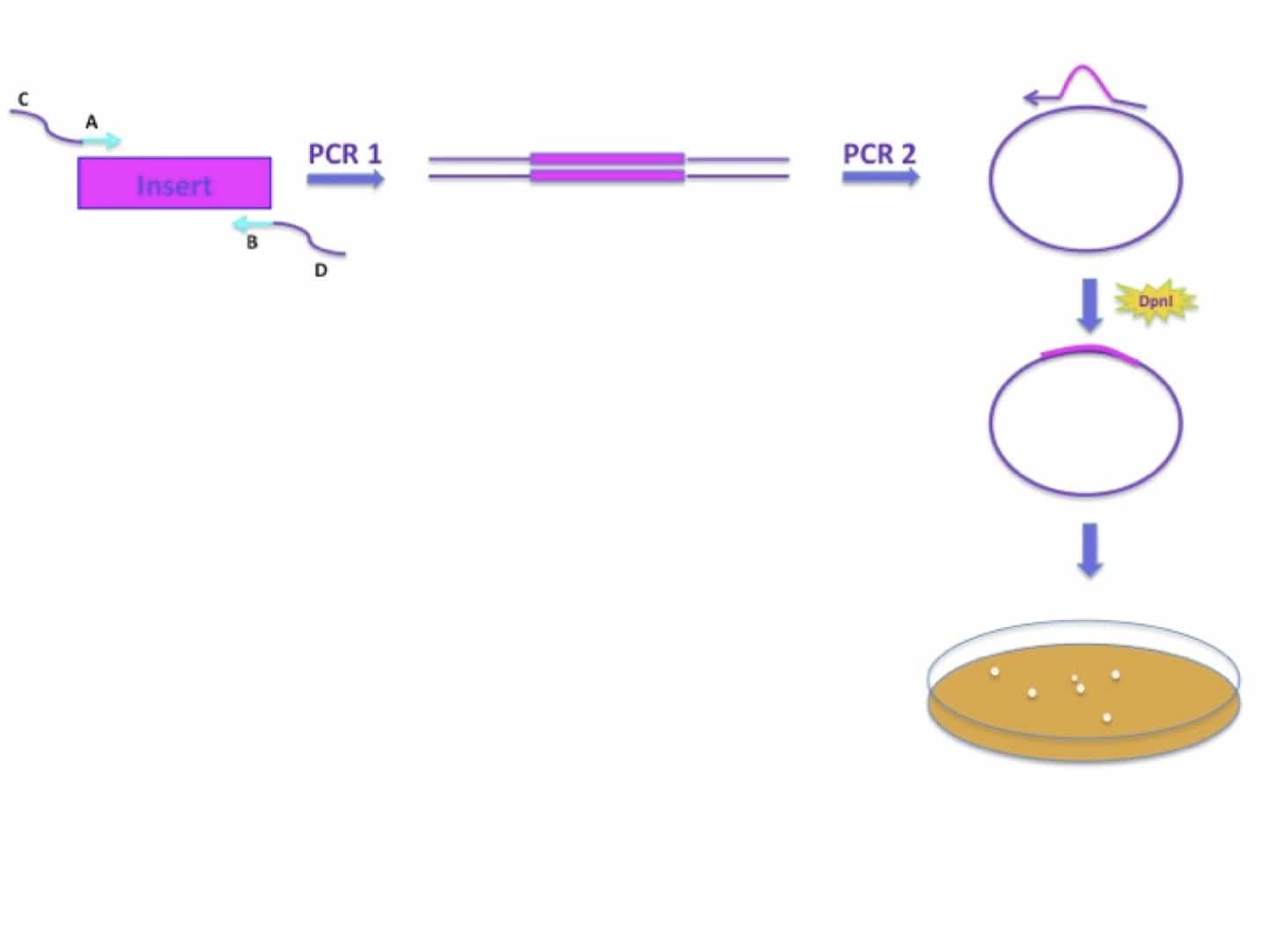
In the past two decade, advances in phage display and antibody engineering have led to the development of phage-displayed antibody technology. 
The advantages of recombinant antibodies are several folds, (i) antibodies can be produced in bacteria, yeast or plant, (ii) immunization is not required and (iii) intrinsic properties such as immunogenicity, affinity, specificity and stability of antibodies can be improved by various mutagenesis technologies. Since the advent of antibody technology, antibody production has moved from hybridoma technology to recombinant DNA methodology. Monoclonal antibodies have become important tools in several fields, including molecular biology, pharmaceutical and medical research, as well as in the treatment of diseases such as cancer and infectious diseases. This advanced method could be adopted by any molecular biology laboratory to generate both naïve or immunized libraries for particular targets as well as for high-throughput applications. These results suggested that by using an optimized protocol and very high repertoire diversity, a compact and efficient phage antibody library can be generated. In particular, we were able to use standard bio-panning method to isolate antibody that can bind to soluble Aflatoxin B1, when using BSA-conjugated toxin as a target, as demonstrated by inhibition ELISA. These included pure recombinant proteins, a hapten and complex antigens such as viral coat proteins, crude snake venom and cancer cell surface antigens. A wide variety of antigens were successfully used to affinity select specific binders. The key difference from other similar libraries was the highest diversity of variable gene repertoires, which was derived from 140 non-immunized human donors. All possible combinations of heavy and light chains, among all immunoglobulin isotypes, were included by using a mixture of primers and overlapping extension PCR. The least number of oligonucleotide primers, electroporations and ligation reactions were used to generate a library of 1.5 × 10 8 individual clones, without generation of sub-libraries. Here we report the simplest and highly efficient method for the construction of a highly useful human single chain variable fragment (scFv) library. However, since this technology emerged more than 20 years ago, there have only been a handful reports on the construction and application of phage display antibody libraries world-wide. In the early days, it was postulated that this technique would eventually replace hybridoma technology and animal immunisations. Phage display technology is a powerful new tool for making antibodies outside the immune system, thus avoiding the use of experimental animals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
